Solar Method to Convert Plastic into Vinegar
| General Studies Paper III: Scientific Discoveries, Environmental Pollution and Degradation |
Why in News?
Recently, researchers at the University of Waterloo developed a solar-powered method to convert plastic waste into acetic acid, the primary component of vinegar, offering a sustainable way to transform plastic pollution into valuable chemicals while reducing environmental damage.
Highlights of the Plastic-to-Vinegar Scientific Breakthrough
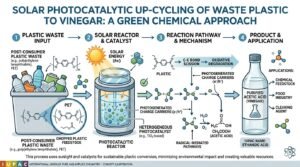
- Discovery: Scientists at the University of Waterloo (Canada) developed a method to convert plastic waste into acetic acid, using sunlight as the primary energy source.
- The research was led by Prof. Yimin Wu and PhD researcher Wei Wei from the Department of Mechanical and Mechatronics Engineering.
- The findings were published in the scientific journal Advanced Energy Materials, highlighting the method as a promising innovation.
- Solar Energy: The technology relies on free solar energy instead of fossil fuels or electricity. Sunlight activates the catalyst and drives chemical reactions that convert plastic molecules into acetic acid.
- Conversion Process: The system uses bio-inspired cascade photocatalysis, mimicking how fungi break down organic matter.
- In the system, plastic waste is mixed in water with the iron-based catalyst and small amounts of hydrogen peroxide.
- Sunlight generates hydroxyl radicals that break plastic chains into smaller fragments, which are then converted into acetic acid (CH₃COOH) through further catalytic reactions.
- Plastics Converted: Experiments showed the method can convert common plastics such as PVC (polyvinyl chloride), PP (polypropylene), PE (polyethylene), and PET (polyethylene terephthalate) into acetic acid. Importantly, it also works with mixed plastic waste, making it practical for real-world recycling systems.
- Laboratory Efficiency: Laboratory tests reported acetic acid production rates of about 63.8 mg·h⁻¹·g⁻¹ from PVC, 12.7 from PE, and around 5 mg·h⁻¹·g⁻¹ from PET and PP under controlled conditions. Even under natural sunlight (~0.6 sun intensity), the system still produced measurable yields.
- Current Stage: The technology is currently at the laboratory research stage, but scientists believe improvements in catalyst design and reactor engineering could enable large-scale solar-powered recycling systems in the future.
What is Vinegar?
|
Significance of this Scientific Breakthrough
- Reducing Global Plastic Waste: The discovery offers a new pathway to address the global plastic pollution crisis, as the world generates about 400 million tonnes of plastic waste annually according to the United Nations Environment Programme (UNEP). Much of this waste ends up in landfills or oceans. By converting plastics into acetic acid, the technology transforms discarded materials into useful chemicals, reducing the volume of waste requiring disposal.
- Alternative to Conventional Recycling: Traditional recycling processes often require high temperatures (above 400°C) and large amounts of energy, while incineration releases carbon dioxide and toxic emissions. The new method relies on solar-driven photocatalysis, significantly lowers energy consumption and emissions, making it a low-carbon recycling solution compared with chemical plastic treatment technologies.
- Mitigating Marine and Microplastic Pollution: Every year, around 11 million tonnes of plastic enter oceans, threatening marine ecosystems and human health. The solar-powered catalytic process works in water, allowing plastics or microplastics to break down into smaller molecules and eventually into acetic acid. This capability creates the potential for technologies that can reduce plastic contamination in aquatic environments in the future.
- Production of Valuable Industrial Chemical: The process produces acetic acid (CH₃COOH), a high-value chemical used in food preservation, pharmaceuticals, textiles, and plastics manufacturing. According to the International Energy Agency, global acetic acid demand exceeds 18 million tonnes annually. Converting plastic waste into this chemical can turn environmental waste into a valuable industrial resource.
- Economic Opportunities in Waste-to-Chemical Industry: Plastic waste management costs governments billions of dollars every year. Technologies that convert waste into chemicals could create a waste-to-value industry where plastics become feedstock for chemical manufacturing. This could generate new industrial markets.
Major Developments in Plastic Waste Conversion
- Enzyme-Based Plastic Breakdown: Scientists at the University of Portsmouth discovered a powerful enzyme called PETase in 2018 that can break down PET plastic used in bottles. Laboratory tests showed the enzyme could degrade PET within a few days, compared with hundreds of years required in nature.
- Super Enzyme Technology: Researchers from University of Portsmouth and the U.S. Department of Energy National Renewable Energy Laboratory developed an enhanced “super enzyme” combining PETase and MHETase. Experiments demonstrated the enzyme could break down PET six times faster than earlier versions.
- Plastic to Jet Fuel Conversion: Scientists at the Washington State University developed a catalytic process that converts polyethylene (PE) plastic waste into jet fuel and lubricants. The method achieved conversion efficiency of about 90%, producing usable hydrocarbon fuels.
- Plastic to Hydrogen Fuel: Researchers from Swansea University and the University of Cambridge created a process that converts plastic waste into hydrogen gas using photocatalysts and sunlight. Hydrogen produced from this reaction can serve as a clean energy fuel.
- Plastic to Vanillin Chemical: Scientists at University of Edinburgh used genetically engineered bacteria to convert PET plastic waste into vanillin, a compound widely used in food flavoring and cosmetics.
- Plastic to Carbon Nanotubes: Scientists at Rice University created a technique called flash Joule heating, which converts plastic waste into graphene and carbon nanotubes within milliseconds. These materials are valuable for electronics, batteries, and advanced composites.
| Also Read: Second Blue Talks Held in India |